Guangshun Wang1, Chuang Han1, Yawen Wang1, Chunliu Yang1, Janette Vardy2,3, Lixin Ma1
1. School of Public Health, Hebei University, Baoding, Hebei Province, China; 2. Sydney Medical School, University of Sydney, Sydney, Australia; 3. Concord Cancer Centre, Concord Hospital, Sydney, Australia. Corresponding to: Lixin Ma, School of Public Health, Hebei University, Baoding, Hebei Province, China, mlx@hbu.edu.cn
Care Weekly 2020;4:1-7
Published online April 17, 2020, http://dx.doi.org/10.14283/cw.2020.1
Abstract
Objective: We performed a systematic review and meta-analysis of cross sectional studies on measuring normal hip BMD.
Methods: The existing studies were conducted in mainland China from the year of 1993 to 2018. Participants were either recruited by population sampling, referred for BMD assessment at routine health checkup or volunteers. The outcomes were hip BMD at ROI of femoral neck and total hip. Methodological qualities were assessed using AHRQ cross-sectional study quality assessment scales. Meta-analysis was conducted applying RevMan software.
Results: 78 cross-sectional studies were retrieved concerning application of six types of DXA scanners in measuring normal hip BMD. The existing studies had reporting bias, participant selection bias and measurement bias. Meta-analysis was made only on two studies which had quality scores of 6. Female Chinese Han had significantly lower femoral BMD values than US Caucasian women standard database from NHANES III (2005-2008) (P < 0.05). The planned subgroup analysis by 5-year of age shows that there are heterogeneities of femoral mean BMD values in age groups of 60~ 69 and over in men and 40~ 49 and over in women (P < 0.1).
Conclusions: Chinese people have a significantly lower normal bone mineral density compared with US Caucasians and the hip BMD losses are distinctive after age of 60 years in men and 40 in women. Our study suggests high quality population-based longitudinal cohort studies on measuring normal hip BMD in future in China.
Keywords: Dual energy X-Ray absorptiometry, cross-sectional study, bone mineral density, hip bone.
Introduction
The diminution of hip bone mass or even osteoporosis affects a significant proportion of aged population worldwide. By 2018, China comprised 1/5 of the world’s population, of whom 12% (166.6/1395.4, in million) are aged ≥65 years. The population aging process is onto China, catching up with developed countries (1). In 2006 osteoporosis potentially affects 69,440,000 mainland Chinese people. In particular, it is estimated that 1 out of 9 women and 1 out of 20 men in the >50 population were affected by this disease, and resulted in 687,000 osteoporosis-related hip fracture in China, accounting for 42% of total number 1,627,000 worldwide in the same periods (2). There was reported that in 2010 osteoporotic fractures led to a costs of approximately 10 billion US dollars to the Chinese healthcare system. And this number and costs will grow to about 6 million fractures costing $25.4 billion annually by the year 2050 (3).
Fragility fractures represent a clinical phase in the natural history of osteoporosis, as they also undermine a patient’s quality of life while burdening the health system. Such consequences including increased disability, social isolation, even partial or complete loss of autonomy in daily activities (4), brought about lose of 5.8 to 7.8 disability adjusted life years (DALYs) in the lifetime course per patient on average (5). And expenses related both to surgical treatment and rehabilitation, were estimated to be about $2733 to $5747 for a fragility fracture patients in 2013 in China (6).
Despite its prevalence (in >50) and high economic and social burden, osteoporosis is perceived as a less severe disease, with respect to cancer and acute myocardial infarction, both by the public and primary care physicians in China. Because of dietary pattern, lifestyle and behavioral risk factors, osteoporosis has become a serious social and public health problem in China (7).
Efficient and accurate diagnostic constitute a fundamental support to clinical practice. For bone fracture prediction and the bone health screening, BMD (bone mineral density) or non-BMD measurements using DXA (Dual energy X-ray absorptiometry) instrument are popular in health care centers (8, 9). Up until now, many small sample size cross-sectional studies measuring normal femoral BMD have been available for Chinese people. To improve the quality of BMD measurement and fracture risk assessment and ultimately improve patient care, we performed a systematic review and meta-analysis on the methodological qualities as well as the effect size of the cross-sectional studies.
Method and materials
Study selection and search strategy
Authors first identified the citations by searching the following electronic databases from inception to July 2018: PubMed, Cochrane Library, Chinese National Knowledge Infrastructure (CNKI, 1979–), Chinese Biomedical Literature Database (CBM, 1978), WanFang database (1982–), and Chinese Scientific Journal Database (VIP, 1989–). The reviewers independently performed the screening of studies, selection, validation, data extraction, and assessment of methodological quality. Disagreements were resolved by consensus with the third author. We selected cross-sectional designed studies that investigated the normal femoral BMD value. The following search terms were used individually and in combination: “femoral”, “femur”, “hip”, “neck”, “trochanter”, “Ward’s”, “bone mineral density”, “bone density”, “normal reference range”, “normal reference value”, “Chinese”, “dual-energy X-ray absorptiometry”, “DEXA”, “DXA”, and “cross-sectional study”.
Inclusion criterion
Citations had to meet the following inclusion criteria: (1) cross-sectional study; (2) participants were Chinese Han ethnicity, ambulatory, living in China, in good health, with no medical complications or receiving treatment for conditions known to affect bone metabolism, including liver or renal failure, malignant tumor, hematological system diseases, rheumatological diseases, hyperthyroidism, diabetes mellitus, primary hyperparathyroidism, bilateral orchidectomy, pituitary or adrenal diseases; (3) outcome were femoral BMD (dividing the amount of bone mineral contents by the area measured, g/cm2) at hip ROI (Region of interest) measured using any type of central DXA instruments, and the hip ROI were defined (8) including: use femoral neck, or total proximal femur whichever the precision error is lowest; BMD may be measured at either hip. Female participants who were pregnant or lactating were excluded. There was no limitation for gender, occupation, educational level etc., except athletes were excluded.
Risk of bias (methodological quality) assessment
The methodological qualities and risk of biases of existing studies were assessed by two authors independently using the 9 items from scale of “AHRQ cross-sectional/prevalence study quality assessment forms” (10). Then risk bias scores were calculated with each item scored 1 point. Trials that met all the above criteria in regard to the impact on the BMD were judged as having a low risk of bias; trials which met none of the criteria were judged as having a high risk of bias and could not be recommended. Trials with insufficient information to classify were regarded as having an unclear risk of bias. Disagreements were resolved by discussion and consensus was made with involvement of the third author where necessary. We choose studies whose risk bias scores are ≥ 6 points for meta-analysis on the hip BMD.
Data analysis
Extractions of data were conducted by the reviewers independently using Microsoft Excel. The structured data extraction forms consisted of bibliographic information, type of DXA scanner, outcomes and measurements, risk of biases and so on. Meta-analysis was performed using RevMan 5.3 software and the generic inverse variance meta-analysis for non-comparative studies was adopted to estimate the effect size of mean BMD (11). Estimated mean femoral BMD (g/cm2) values at ROI were pooled in specific age, gender, types of DXA scanners. The effect size was presented as an estimation of mean with 95% confidence intervals (CIs). A fixed effects model was used unless there was evidence of heterogeneity. We assessed heterogeneity using the chi-squared test and/or I-squared statistic. We considered an α ≤ 0.1 and/or I2 ≥50 % was indicative of substantial heterogeneity. When heterogeneity was present, subgroup analysis was conducted according to planned age interval of 5-year. Then based on interval estimation and statistical inference of population mean, we made a comparison between BMD levels of Chinese and that of US Caucasians taken from the database NHANES III (the third national health and nutrition examination survey) 2005-2008 (11) to elucidate trends of BMD changes between the two races.
Results
Description of studies
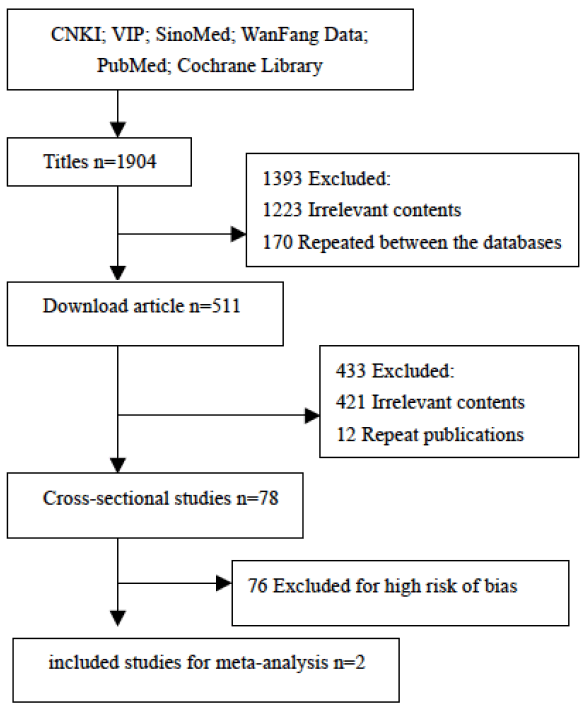
Figure 1 outline the citation searching process and study selection. A total of 78 cross-sectional studies (N = 108,392) published from 1993 to 2018 were retrieved. 76 articles were published in full in Chinese and two in English.
Participants were aged from 2 to 102 years old, and were from 23 administrative provinces or municipalities throughout China. Their femoral neck and total hip BMD were measured using central DXA systems available in their local hospital. The following types of DXA were used in the 78 studies: GE Lunar (42/78, 53%); Hologic (19/78, 24%); Norland (6/78, 7%); DMS Challenger (9/78, 11%); Medlink Osteocore (2/78, 3%); and I’ACN (2/78, 3%) (Two types of scanners were used in one study). The investigation intervals varied from 0.4 to 13.33 years with a median of 3 years. Femoral BMD were measured at region of femoral neck (75/78, 96%), total hip (11/78, 14%), trochanter (70/78, 90%), inter-trochanter (5/78, 6%), and Ward’s triangle (Ward’s) (68/78, 87%) respectively.
Methodological qualities of the existing studies
The qualities of original studies were assessed as following, see Table 1. (1) The source of information of the existing studies was collected through cross-sectional studies in mainland Chinese Han. Of these, 77% (60/78) were clinical record reviews on routine health checkup; 14% (11/78) were surveys recruiting subjects based on population sampling; and 9% (7/78) indicated their subjects were volunteers. None of the studies reported a flow chart or a clinical trial identifier number. (2) Approximately 91% (71/78) of the studies listed inclusion and exclusion criteria for subjects, others only referred to ‘normal people’. (3) Roughly less than a half of the studies (33/78, 42%) did not indicate the time period used for identifying subjects. (4) Overall, 24% (19/78) of the studies reported the radiologists of the BMD measurement were trained before investigation, and 54% (36/78) referred that quality control of the measurement was accomplished through periodical calibration on DXA instruments using anthropomorphic phantoms at its own health care center. (5) Approximately half (40/78, 51%) of the studies described the precision of test/retest of BMD measurements for quality assurance purposes. (6) About one fifth (17/78, 22%) of the studies described the proportion of the baseline confounding factors such as body weight, height, menstruation status, diet and eating habits. (7) None of the studies explained if any subjects were excluded from the analysis or how missing data were handled. In conclusion, 81% (63/78) of the original studies had risk bias scores of less than 5, and less than 2% (2/78) had scores of 6 (12, 13).
Note: a) AHRQ: The Agency for Healthcare Research and Quality; Note: a) AHRQ: The Agency for Healthcare Research and Quality; The 5th and 11th item are no relevent to the risk bias assessment; The 5th and 11th item are no relevent to the risk bias assessment; (5) Indicate if evaluators of subjective components of study were masked to other aspects of the status of the participants; (5) Indicate if evaluators of subjective components of study were masked to other aspects of the status of the participants; (11) Clarify what follow-up, if any, was expected and the percentage of patients for which incomplete data or follow-up was obtained; (11) Clarify what follow-up, if any, was expected and the percentage of patients for which incomplete data or follow-up was obtained
Pooling results of hip BMD
Estimation of population means of hip BMD and its 95% confidence intervals
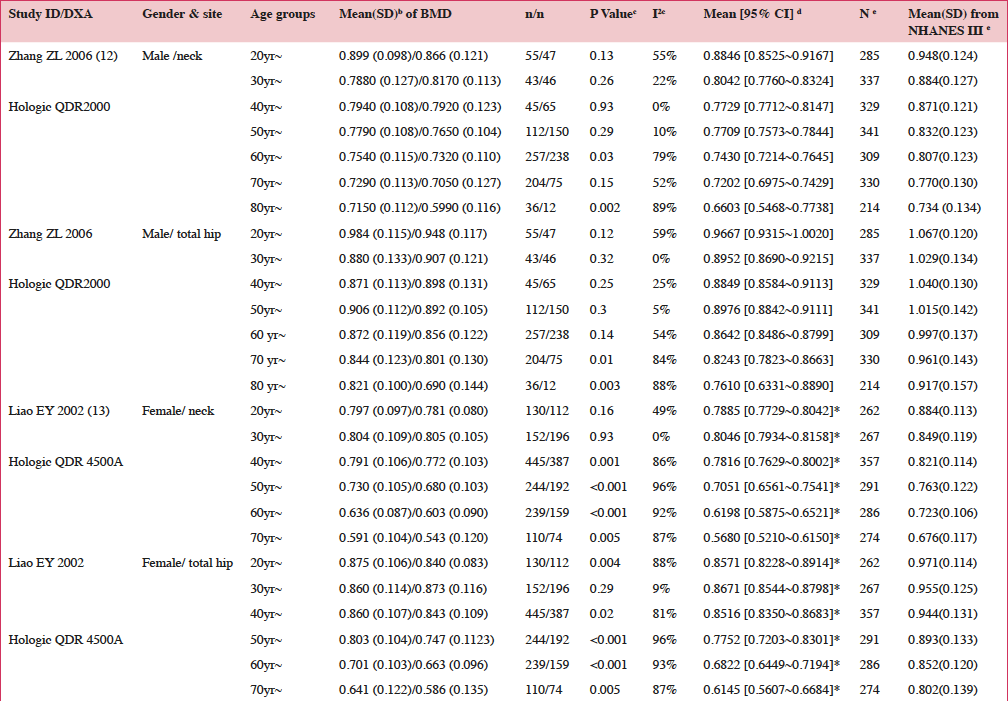
Among the 78 cross-sectional studies, only two surveys (12, 13) met the conditions for meta-analysis were included. Table 2 outlines the femoral BMD levels, pooling results and US Caucasians reference database. Highlight that normal bone losses are age related. The peak total hip BMD appears to be reached in the 20 ~ 29 years age group both in men and women respectively (female neck BMD appears highest at 30 ~ 39 age group). Subsequently they begin to lose mineral mass in the 40 ~ 49 year age group. Women from the age of 40 ~ 49 showed a faster rate of decline in mean BMD than men matched by age- group.
Notes: b. Mean(SD) of femoral BMD classified by age intervals of 5-year for Chinese people scanned by Hologic QDR 4500A; c. Heterogeneity test across the Mean (SD) of femoral BMD; d. Pooling result of Mean of BMD; e. Reference data reported from NHANES III (2005-2008) for Non-Hispanic white scanned by Hologic QDR 4500C; * P < 0.05 comparison between pooling result and the reference data
Subgroup analysis
Subgroup analysis of femoral BMD was explored by planned age interval of 5-year. There were heterogeneities of femoral mean BMD values in age group of 40~49 and over in women, and 60~69 and over in men (P < 0.1), See Table 2.
Comparison of mean femoral BMD (g/cm2) value and statistical inference
Table 2 shows the comparison of female mean hip BMD in categories of Hologic QDR 4500 between female Chinese and US Caucasians Standard Database of NHANES III (3). The Chinese normative BMD data partially overlaps head and tail with Caucasian BMD values. Female US Caucasians had a statistical significant higher femoral BMD value than Chinese women (9, 12-13) (P < 0.05).
Discussion
The societal and economic burden of osteoporosis are increasing as the population ages in China, as in other developing countries. BMD measurement and fracture risk prediction will continue to be needed. In our review, a variety of different central DXA instruments and techniques are applied by radiologists in different clinical settings in china. However due to practical limitations in the surveys such as time, budget, the process of measurement and outcome reporting of hip BMD in some studies might not met the official position of ISCD 2015 (The International Society for Clinical Densitometry) (8). Our review shows that the following methodological qualities need to be improved in future studies.
Methodological quality of the existing studies
There have been a number of methodological challenges to the majority of the original studies. (1) Reporting bias: this occurred when missing or inexplicit reporting some items relevant to cross-sectional survey on BMD measurement. These items are including baseline information (demographic and anthropometric data), flow chart of clinical studies, missing data and statistical disposal methods. They are valuable in reading the procedure of the original studies and in assessing its internal and external validities. And they are also critical in developing best clinical practices in the acquisition, interpretation, and clinical application of normal BMD. Additionally, we found that some studies selected reporting normal hip BMD at regions of Wards’ or other anatomic site rather than that at ROIs of femoral neck or total hip. These areas are not sensitive in predicting fracture risk based on ISCD official positions (8). (2) Participant selection bias: few of the existing studies reported that their sample was population-based, or mentioned consecutive subjects of clinical record review, so it was difficult to judge the integrity of data collection. Some study had small sample sizes (less than 30), which increases the likelihood of sampling error. Furthermore, their results of BMD have poor representativeness of the studied population. Small sample size in some groups also have lower statistical power, which could enlarge the width of normal mean hip BMD distribution leading to false negative statistical inference in diagnosis of osteoporosis. (3) Measurement bias: one half of the studies did not report key measurement parameters of precision or LSC, making it is impossible to differentiate whether risk bias was introduced by the instrument or radiologist in BMD measurements or the data had systematic deviation. Failure to comply with manufacturers’ recommendations for routine device maintenance and quality control also might result in unreliable BMD measurements. Issues such as calibration shifts can also occur after moving a DXA system, following reassembly or breakage of its components. Regular measurement of a phantom will detect these changes and render the device can be recalibrated in time (8, 9). In a word, we recommend future researchers follow the official position from ISCD on measuring normal hip BMD (8) and the STROBE statement (Strengthening the Reporting of Observational Studies in Epidemiology) in reporting cross-sectional /prevalence study. Our study certainly advocate training programs for DXA radiologists to improving the quality on evaluating hip BMD and certification program to encourage technologists and radiologists to keep their knowledge and skills current.
Trends of BMD changes and the comparison of hip BMD between Chinese and Caucasians
Our study shows bone diminutions in the hip begin relatively early in life, in the age group of 40 ~ 49 years in Chinese women and men. First, these BMD changes are in general agreement with the trends of J C Stevenson (14) who stated that the peak femoral bone mineral density of both male and female Caucasians occurs around 30 ~ 35 years of age. Second, Chinese Han women have a statistically significant lower hip BMD level than age- matched American Caucasian women from previous research (9, 15). This might mainly due to the difference of their dietary calcium intakes and different food consumption patterns (8). A systematic review (16) on dietary calcium intakes from the year of 2000 to 2007 found that the mean calcium intakes were 450.4 mg/day for average Chinese adult, which were about half of dietary calcium assumption of 912 ± 217 mg/day for middle adults living in all parts of England from 1982 to 1989 (17). Except for lower calcium intake, environmental pollutions and sedentary lifestyle may also play a role for relatively lower level of hip bone mass for Chinese people (18, 19).
Our review also advise that normal hip BMD levels might be classified by age interval of equal or less than 5-year for participants of 40 years and over in women and 60 and over in men in future studies. Considering the needs of long-term surveillance and assessment on the changes of BMD and risk factors, a design of population-based randomized longitudinal cohort perspective study rather than cross-sectional studies would be recommended in future study in clinical practice (20).
Conclusion
Chinese people have a significantly lower normal bone mineral density compared with US Caucasians and hip BMD losses are distinctive after age of 60 years in men and 40 in women. A well designed, national wide population-based longitudinal prospective cohort study on measuring normal hip BMD is recommended in China. And tremendous efforts should pay for high-quality education programs for technologists and radiologists to assure their knowledge and competence, and sound methodology in BMD measurement to develop and update scientifically grounded best clinical practices in management of osteoporosis.
Funding
The sponsors had no role in the review or approval of the manuscript.
Acknowledgments
This work was supported by the Key medical research project from the Department of Health of Hebei Province (No.07130 to Lixin Ma); Hebei University Natural Science Foundation (2013-264, and 2007Y04 to Lixin Ma); Project of innovation and scientific research training for Hebei University students (No. 2018168 to Lixin Ma and Jing Liu, etc); and Project of innovation and scientific research training for college students of Hebei Province (No.201710075084 to Chunliu Yang and Lixin Ma); We would like to thank all participants and investigators in this study.
Conflicts of Interest
The authors have declared no conflicts of interest.
References
1. Editorial Board of Osteoporosis Prevention and Treatment from the China Health Promotion Foundation. China white paper on osteoporosis. Chinese Journal of Health Management 2009; 3(3):148-154. DOI:10.3760/cma.j.issn.1674-0815.2009.03.006.
2. Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 2006; 17(2):1726-1733. DOI 10.1007/s00198-006-0172-4(8).
3. Si L, Winzenberg TM, Jiang Q, Chen M, Palmer AJ. Projection of osteoporosis-related fractures and costs in China: 2010–2050. Osteoporos Int 2015; 26(7): 1929-1937. Doi:10.1007/s00198-015-3093-2.
4. Watts NB, Bilezikian JP, Camacho PM, Greenspan SL, Harris ST, Hodgson SF, et al. American association of clinical endocrinologists medical guidelines for clinical practice for the diagnosis and treatment of postmenopausal osteoporosis: executive summary of recommendations. Endocrine Practice 2010; 16(6): 1016. doi:10.4158/EP.16.6.1016
5. Josep Darbà, Lisette Kaskens, Nuria Pérez-Álvarez, Santiago Palacios, José Luis Neyro, Javier Rejas. Disability-adjusted-life-years losses in postmenopausal women with osteoporosis: a burden of illness study. BMC Public Health 2015; 15(1): 324. doi: 10.1186/s12889-015-1684-7
6. Yang Y, Du F, Lu T, Xie Z, Xu W, Huang Q, Ye WW, Burge R. Outpatient medical costs, indirect costs, and family burden of osteoporosis fracture in China. Value in Health 2013; 16(7): A561. doi:10.1016/j.jval.2013.08.1481
7. Liu Z, Pang SJ, Song Pk, et al. Dietary calcium intake among Chinese elderly during 2010-2012. Acta Nutrimenta Sinica 2017; 39(5): 442-447.
8. Shepherd JA, Schousboe JT, Broy SB, et al. Executive Summary of the 2015 ISCD position development conference on advanced measures from DXA and QCT: fracture prediction beyond BMD. J Clin Densitom 2015; 18(3): 274-286.
9. Looker AC, Borrud LG, Hughes JP, et al. Lumbar spine and proximal femur bone mineral density, bone mineral content, and bone area: United States, 2005–2008. National Center for Health Statistics. Vital Health Stat 11(251). 2012.
10. Rostom A, Dubé C, Cranney A, et al. Celiac Disease. Rockville (MD): Agency for Healthcare Research and Quality (US); 2004 Sep. (Evidence Reports/Technology Assessments, No. 104.) Doi: https://www.ncbi.nlm.nih.gov/books/NBK35149/. Accessed 7 June 2019.
11. Jessica S, Wang Memoli, Paul J Nietert, Gerard A Silvestri. Meta-analysis of guided bronchoscopy for the evaluation of the pulmonary nodule. Chest 2012;142(2):385-393.
12. Zhang ZL, Qin YJ, Huang QR, et al. Bone mineral density of the spine and femur in healthy Chinese men. Asian J Androl 2006;8(4):419-427.
13. Liao EY, Wu XP, Deng XG, et al. A cross-sectional survey of bone mineral density reference values at multiple skeletal sites in women from Changsha area in China. Chin J Osteoporos 2002;8(2):97-101.
14. Stevenson JC, Lees B, Devenport M, et al. Determinants of bone density in normal women: risk factors for future osteoporosis? Br Med J 1989;298:924-928.
15. Walker MD, Babbar R, Opotowsky AR, et al. A referent bone mineral density database for Chinese American women. Osteoporos Int 2006;17:878-887.
16. Dietary Calcium Intake from 2000 to 2007. Prev Med Trib 2009;15(7): 577-583.
17. Price GM, Paul AA, Key FB, et al. Measurement of diet in a large national survey: comparison of computerized and manual coding of records in household measures. J Hum Nutr Diet 1995; 8:417-428.
18. Kanis JA. Assessment of osteoporosis at the primary health-care level. WHO Collaborating Center for Metabolic Bone Diseases. Technical Report. University of Sheffield, UK; 2007. https://www.shef.ac.uk/FRAX/pdfs/WHO_Technical_Report.pdf. Accessed 7 June 2019.
19. Barbara R, Ann L. Ann LG, Gregory H, et al. Measurement of function post hip fracture: testing a comprehensive measurement model of physical function. Rehabil Nurs 2016; 41(4): 230-247.
20. Breslow NE, Day NE. Statistical Methods in cancer research Volume Ⅱ-The design and analysis of cohort studies. Lyon: International Agency for research on cancer; 1987.